Estimation of species divergence time
- Orthfiner2 example is OrthoFinder v2.5.4
- PAML example is paml4.10.6
- Muscle v5 example is Muscle v5.1
- Biopython or Biopython.cn
1.coding sequence of each species in fasta format in one folder
 2.protein sequence of each species in fasta format in one folder
2.protein sequence of each species in fasta format in one folder
 3.Merge all cds files into the your_working directory
3.Merge all cds files into the your_working directory
cat cds_in_each_species/*.fa > your_working_directory/sol.cds.all.faUse the protein foler of the species as input to execute the orthfinder software. The prefix of each cds file is species identification, and the recommended suffix is fa, which is convenient for subsequent processes to read.
./orthofinder -f pep_in_each_species/Add or remove species can be combined with-b and-f parameters, refer to the instructions for the use of orthfinder(Orthfiner2 readme)
Copy useful orthfiner results(Orthogroups_SingleCopyOrthologues.txt, Orthogroups.txt and SpeciesTree_rooted.txt) to the working directory
cp Orthogroups/Orthogroups_SingleCopyOrthologues.txt Orthogroups/Orthogroups.txt Species_Tree/SpeciesTree_rooted.txt workDirectoryReferring to the phylogenetic tree of orthofinder and the fossil time range of Timetree, rewrite the tree file
View and preprocess the tree of orthofinder
3.1.1 check tree file
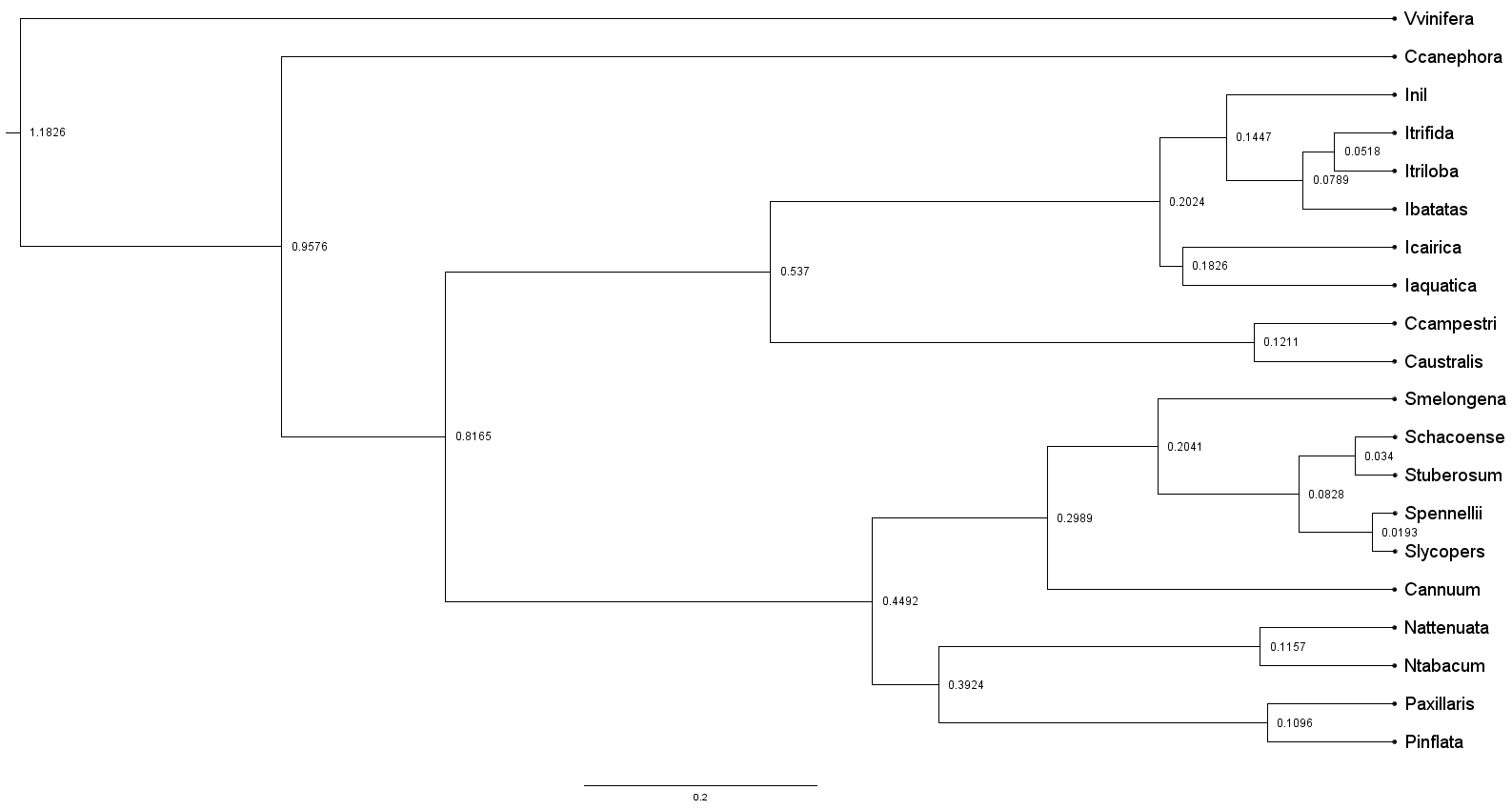
cat SpeciesTree_rooted.txt(Vitis_vinifera:0.124147,(Coffea_canephora:0.196146,(Solanum_lycopersicum:0.195935,((Cuscuta_campestris:0.0146811,Cuscuta_australis:0.0347711)0.949219:0.185453,((Ipomoea_nil:0.0217926,Ipomoea_aquatica:0.0289792)0.253723:0.0123219,(Ipomoea_batatas:0.387199,(Ipomoea_triloba:0.00613322,Ipomoea_trifida:0.0061229)0.421138:0.0184493)0.440876:0.0245531)0.718464:0.0782321)0.78037:0.0732721)0.65297:0.0417202)1:0.124147);3.1.2 pre-processing tree file
python phyTree.py -c SpeciesTree_rooted.txt SpeciesTree_rooted.rewrite.txtcat SpeciesTree_rooted.rewrite.txt(Vvinifera,(Ccanephora,((((Inil,((Itrifida,Itriloba),Ibatatas)),(Icairica,Iaquatica)),(Ccampestri,Caustralis)),(((Smelongena,((Schacoense,Stuberosum),(Spennellii,Slycopers))),Cannuum),((Nattenuata,Ntabacum),(Paxillaris,Pinflata))))));Remember to change the species name to less than 10 characters
vi SpeciesTree_rooted.rewrite.txt 20 1
(Vvinifera,(Ccanephora,((((Inil,((Itrifida,Itriloba),Ibatatas)),(Icairica,Iaquatica)),(Ccampestri,Caustralis)),(((Smelongena,((Schacoense,Stuberosum),(Spennellii,Slycopers))),Cannuum),((Nattenuata,Ntabacum),(Paxillaris,Pinflata))))));20 and 1 represent the number of species and trees, respectively
3.3 time search in Timetree and reconstituted species tree
3.3.1 Query the existing fossil time
The divergence time between Coffea canephora and Solanum lycopersicum is about 72.4-104.9mya.
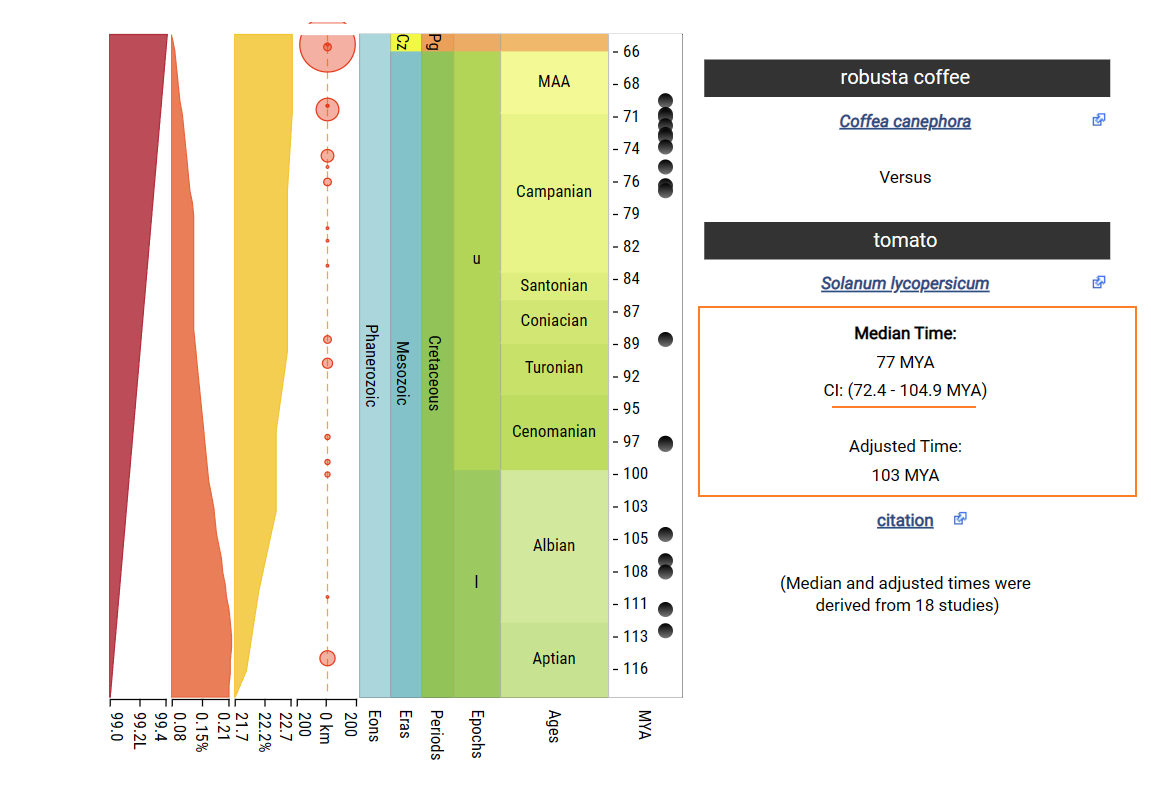
The divergence time between Coffea canephora and Solanum lycopersicum is about 112.4-125.0mya.
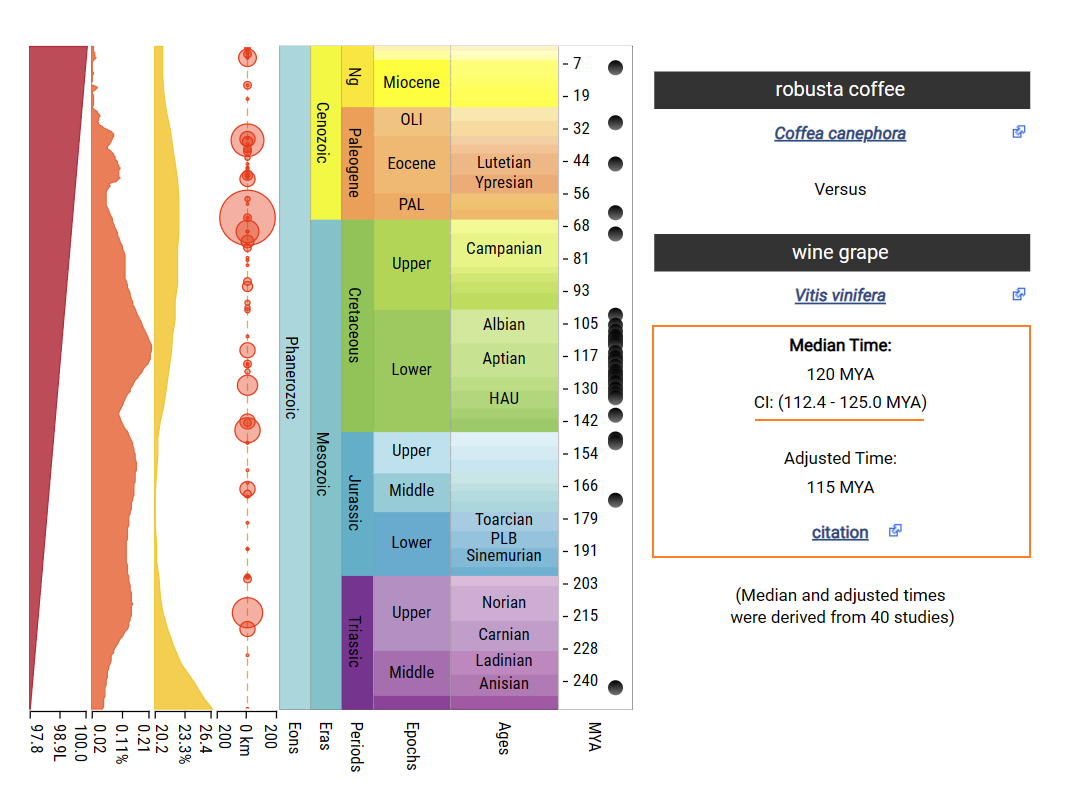
3.3.2 Add calibration time to tree file
vi SpeciesTree_rooted.rewrite.txt 20 1
(Vvinifera,(Ccanephora,((((Inil,((Itrifida,Itriloba),Ibatatas)),(Icairica,Iaquatica)),(Ccampestri,Caustralis)),(((Smelongena,((Schacoense,Stuberosum),(Spennellii,Slycopers))),Cannuum),((Nattenuata,Ntabacum),(Paxillaris,Pinflata)))))'>0.724<1.049')'>1.124<1.25';
//end of file4.1 View script help
python singleCopyAlign2paml_workflow.py -husage: extractSingleCopySequence.py [-h] oscgroups ogroups allcds
用途:提取Orthogroups.txt中的单拷贝基因序列,并对每组单拷贝基因进行序列比对。
positional arguments:
oscgroups Orthfinder resulte file: "Orthogroups_SingleCopyOrthologues.txt" (type = str)
ogroups Orthfinder resulte file: "Orthogroups.txt" (type = str)
allcds a merged coding squence file of all used species (type = str)
optional arguments:
-h, --help show this help message and exit4.2 Execute script
python singleCopyAlign2paml_workflow.py Orthogroups_SingleCopyOrthologues.txt Orthogroups.txt sol.cds.all.faThis script is equivalent to using the following script in turn:
extractSingleCopySequence.py --> batchId2Spname.py --> batchSeqAlign.py --> fasAlign2phy.py -->delStopCodon.py
Use the -h parameter to understand how to use it.
cp mcmctree.ctl sol.mcmctree.ctl
vi sol.mcmctree.ctl seed = -1
seqfile = mtCDNApri123.txt ### Change to the alignment file name: all.delStopCodon.phys
treefile = mtCDNApri.trees ### Change to the tree file name: SpeciesTree_rooted.rewrite.txt
mcmcfile = mcmc.txt
outfile = out.txt
ndata = 2 ### chang to alignment number,this example is 2
seqtype = 0 * 0: nucleotides; 1:codons; 2:AAs
usedata = 1 * 0: no data; 1:seq like; 2:normal approximation; 3:out.BV (in.BV)
clock = 2 * 1: global clock; 2: independent rates; 3: correlated rates
RootAge = '<1.0' * safe constraint on root age, used if no fossil for root.
model = 0 * 0:JC69, 1:K80, 2:F81, 3:F84, 4:HKY85
alpha = 0 * alpha for gamma rates at sites
ncatG = 5 * No. categories in discrete gamma
cleandata = 0 * remove sites with ambiguity data (1:yes, 0:no)?
BDparas = 1 1 0.1 * birth, death, sampling
kappa_gamma = 6 2 * gamma prior for kappa
alpha_gamma = 1 1 * gamma prior for alpha
rgene_gamma = 2 20 1 * gammaDir prior for rate for genes
sigma2_gamma = 1 10 1 * gammaDir prior for sigma^2 (for clock=2 or 3)
finetune = 1: .1 .1 .1 .1 .1 .1 * auto (0 or 1): times, musigma2, rates, mixing, paras, FossilErr
print = 1 * 0: no mcmc sample; 1: everything except branch rates 2: everything
burnin = 2000
sampfreq = 10
nsample = 20000
*** Note: Make your window wider (100 columns) before running the program.
mcmctree sol.mcmctree.ctlultrametric tree of selected species in NEXUS format(The time standard is 100 million years)
vi FigTree.tre#NEXUS
BEGIN TREES;
UTREE 1 = (Vvinifera: 1.182571, (Ccanephora: 0.957549, ((((Inil: 0.144686, ((Itrifida: 0.051791, Itriloba: 0.051791) [&95%HPD={0.0180103, 0.0907333}]: 0.027058, Ibatatas: 0.078849) [&95%HPD={0.0342785, 0.127052}]: 0.065837) [&95%HPD={0.0765007, 0.224146}]: 0.057742, (Icairica: 0.182582, Iaquatica: 0.182582) [&95%HPD={0.092973, 0.282555}]: 0.019846) [&95%HPD={0.112743, 0.30083}]: 0.334590, (Ccampestri: 0.121145, Caustralis: 0.121145) [&95%HPD={0.037339, 0.218144}]: 0.415872) [&95%HPD={0.368526, 0.701565}]: 0.279506, (((Smelongena: 0.204091, ((Schacoense: 0.034018, Stuberosum: 0.034018) [&95%HPD={0.0081394, 0.0673773}]: 0.048750, (Spennellii: 0.019273, Slycopers: 0.019273) [&95%HPD={0.0033041, 0.0397274}]: 0.063495) [&95%HPD={0.038023, 0.136205}]: 0.121324) [&95%HPD={0.113544, 0.305304}]: 0.094774, Cannuum: 0.298865) [&95%HPD={0.178539, 0.422947}]: 0.150367, ((Nattenuata: 0.115726, Ntabacum: 0.115726) [&95%HPD={0.0393255, 0.212033}]: 0.276721, (Paxillaris: 0.109594, Pinflata: 0.109594) [&95%HPD={0.0401356, 0.193081}]: 0.282852) [&95%HPD={0.244036, 0.546504}]: 0.056785) [&95%HPD={0.302158, 0.609576}]: 0.367292) [&95%HPD={0.652822, 0.961443}]: 0.141026) [&95%HPD={0.794656, 1.06636}]: 0.225022) [&95%HPD={1.12196, 1.24766}];
END;2.Open and convert with Figtree and convert to nwk format
export as a Newick: File > Save Trees > Select 'Newick' from menu.
If you save it and name it FigTree.tre.nwk.
cat FigTree.tre.nwk(Vvinifera:1.182571,(Ccanephora:0.957549,((((Inil:0.144686,((Itrifida:0.051791,Itriloba:0.051791):0.027058,Ibatatas:0.078849):0.065837):0.057742,(Icairica:0.182582,Iaquatica:0.182582):0.019846):0.33459,(Ccampestri:0.121145,Caustralis:0.121145):0.415872):0.279506,(((Smelongena:0.204091,((Schacoense:0.034018,Stuberosum:0.034018):0.04875,(Spennellii:0.019273,Slycopers:0.019273):0.063495):0.121324):0.094774,Cannuum:0.298865):0.150367,((Nattenuata:0.115726,Ntabacum:0.115726):0.276721,(Paxillaris:0.109594,Pinflata:0.109594):0.282852):0.056785):0.367292):0.141026):0.225022);
With a little modification, the above results can be used in the input file of CAFE and CAFE5.
For a typical CAFE analysis, users are most interested in determining two things:
* Which gene families are rapidly evolving * The branches of the tree on which these families are rapidly evolving
1. Modify the Orthogroups.GeneCount.tsv file of OrthoFinder to generate a gene family count file that meets the requirements of CAFE5.
awk 'OFS="\t" {$NF=""; print}' Orthogroups.GeneCount.tsv > tmp && awk '{print "(null)""\t"$0}' tmp > cafe.input.tsv && sed -i '1s/(null)/Desc/g' cafe.input.tsv && rm tmp2.Filtering exception family
After that, it is necessary to eliminate the gene families with excessive copy number differences between different species, otherwise an error will be reported. CAFE5 has built-in scripts to use, and sometimes you need to remove the first line before you run it.
python ~/soft/CAFE5/tutorial/clade_and_size_filter.py -i cafe.input.tsv -o gene_family_filter.txt -s The output should be two CAFE input files, one for gene families with < 100 gene copies in all species(gene_family_filter.txt), another for the remaining gene families(large_gene_family_filter.txt). The first file should be used to estimate parameter values, and these values should then be used to analyse the second file. Refer to the cafetutorial_clade_and_size_filter.py of cafe_tutorial
3. Ultrametric tree in Newick format
The time of the previous Ultrametric tree needs to be multiplied by 100, so the time base becomes MYA, which can be directly used as the second input file.
sed 's/\([),]\)/\n\1/g' FigTree.tre.nwk | awk -F ":" '{if($2~/[0-9]/){printf $1":"$2*100}else{print $0}}'(Vvinifera:118.257,(Ccanephora:95.7549,((((Inil:14.4686,((Itrifida:5.1791,Itriloba:5.1791):2.7058,Ibatatas:7.8849):6.5837):5.7742,(Icairica:18.2582,Iaquatica:18.2582):1.9846):33.459,(Ccampestri:12.1145,Caustralis:12.1145):41.5872):27.9506,(((Smelongena:20.4091,((Schacoense:3.4018,Stuberosum:3.4018):4.875,(Spennellii:1.9273,Slycopers:1.9273):6.3495):12.1324):9.4774,Cannuum:29.8865):15.0367,((Nattenuata:11.5726,Ntabacum:11.5726):27.6721,(Paxillaris:10.9594,Pinflata:10.9594):28.2852):5.6785):36.7292):14.1026):22.5022);4. Run CAFE
4.1 Copy and rename the shell script for cafe
cp ~soft/CAFE/example/cafe_script2.sh your_working_directory/cafe_script_sol.sh4.2 Modify the shell script
Before modify
vi cafe_script_sol.sh#!shell
date
version
#specify data file, p-value threshold, # of threads to use, and log file
load -i example_data.tab -p 0.01 -t 10 -l log.txt
#the phylogenetic tree structure with branch lengths
tree (((chimp:6,human:6):81,(mouse:17,rat:17):70):6,dog:93)
#search for 2 parameter model
lambda -s -t (((2,2)1,(1,1)1)1,1)
# generate a report
report report.txtAfter modify
vi cafe_script_sol.sh#!shell
date
#specify data file, p-value threshold, # of threads to use, and log file
load -i gene_family_filter.txt -p 0.01 -t 10 -l log.txt
#the phylogenetic tree structure with branch lengths
tree (Vvinifera:118.257,(Ccanephora:95.7549,((((Inil:14.4686,((Itrifida:5.1791,Itriloba:5.1791):2.7058,Ibatatas:7.8849):6.5837):5.7742,(Icairica:18.2582,Iaquatica:18.2582):1.9846):33.459,(Ccampestri:12.1145,Caustralis:12.1145):41.5872):27.9506,(((Smelongena:20.4091,((Schacoense:3.4018,Stuberosum:3.4018):4.875,(Spennellii:1.9273,Slycopers:1.9273):6.3495):12.1324):9.4774,Cannuum:29.8865):15.0367,((Nattenuata:11.5726,Ntabacum:11.5726):27.6721,(Paxillaris:10.9594,Pinflata:10.9594):28.2852):5.6785):36.7292):14.1026):22.5022)
#
lambda -s
#search for 2 parameter model
#lambda -s -t (((2,2)1,(1,1)1)1,1)
#specify the global lambda to for generating simulated data
#lambda -l 0.0017
#generate 10 simulated data sets
#genfamily rndtree/rnd -t 10
#estimate lambdas and compare likelihoods of global lambda and 2-parameter models
#lhtest -d rndtree -l 0.0017 -t (((2,2)1,(1,1)1)1,1) -o lh2.out
# report output
report sol_cafe_out
date4.3 Execute cafe
/home/xian/soft/CAFE/release/cafe cafe_script_sol.sh4.4 Integrate cafe results
In the python_scripts directory of cafe_tutorial, there is a script called cafetutorial_report_analysis.py (written by python2) that can collates the results of cafe.
conda activate python27
python cafetutorial_report_analysis.py -i sol_cafe_out.cafe -o sol_cafe_out.summaryAs a result, four files appear, all prefixed with the value of the-o parameter.
-
sol_cafe_out.summary_anc.txt
每个家族在每个节点变化(扩张/收缩)的数量. The number of changes(expansion or contraction) in each family at each node. -
sol_cafe_out.summary_fams.txt 每个节点变化(扩张/收缩)的家族编号(*表示显著). Family IDs that changes(expansion or contraction) at each node(\ * indicates significant).
-
sol_cafe_out.summary_node.txt 每个节点中变化(扩张/收缩)的基因家族数量. The number of gene families that change(expansion or contraction) in each node
-
sol_cafe_out.summary_pub.txt 每个物种中基因家族的变化数量. The number of changes in gene families in each species.
For the parameter settings and the meaning of the results, please read the CAFE documentation in detail.
4.5 CAFE_fig
The results of CAFE can be displayed through CAFE_fig. CAFE_fig requires Python3.4+ and ETE3: Install ETE3 with
pip3 install 'ete3==3.0.0b35'
or
pip3 install 'ete3==3.1.2'python CAFE_fig.py sol_cafe_out.cafe -pb 0.01 -pf 0.01 --dump test -g pdf 5. Run Cafe5
cafe5 -i gene_family_filter.txt -t FigTree.tre.nwk -o outCafe5 uses the Base model by default, and you can use the Gamma model with the-k parameter.
For the parameter settings and the meaning of the results, please read the CAFE5 documentation in detail.
Note1: Well, this small process is equivalent to 2.Preparing the input in cafe5, but it's a little easier to operate.
Note2: There are four parts of the files or folders in the process are not provided in this github directory, namely: ./cds_in_each_species, ./pep_in_each_species, sol.cds.all.fa and OrthoFinder result directory. The reason is that I can't upload large files successfully.
Note3: This is a problem showing the results of CAFE5, because CAFE_fig is no longer available for CAFE5 and can only be used for older versions of CAFE. If I have time later, I will modify the script to make it easy to use..